Pathophysiology of snoring and obstructive sleep apnoea
An increasing number of dentists are managing sleep-related breathing disorders as a result of awareness of the role they can play in improving the quality of life of patients who suffer with this debilitating disease. What are its causes and consequences?
The upper airway in humans has multiple purposes: speech, swallowing and breathing. It comprises soft tissue and muscle, but, importantly in the context of this article, does not have hard, bony tissue to prevent the muscle or soft tissue from compressing or collapsing.
The collapsible part of the upper airway, which spans the hard palate to the larynx, can change shape and close briefly to allow for speech and swallowing. It is the collapse of this upper airway during sleep that precipitates an apnoeac event.
Sleep-disordered breathing
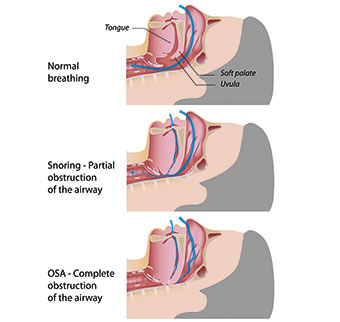
The upper airway collapses during sleep when there is no compensatory input to the motor neurones of the upper airway dilator muscles – mostly the genioglossus and tensor palati muscles. How often the person compensates for these collapses determines the rate at which the cycle repeats. When airflow is blocked for 10 seconds or more, it is called apnoea (or apnea) meaning suspension of breathing. Hypopnoea (or hypopnea) is a partial collapse of the airway that results in an airflow reduction of greater than 50 per cent for 10 seconds or more (Fig 1).
Although several neurotransmitters and neuromodulators have been identified as contributing to the regulation of the upper airway opening, there has been little progress finding a medicine to prevent its collapse.
Burwell, Robin, Waley and Bickelmann studied the pathophysiology of sleep apnoea-hypopnoea syndrome (SAHS) in the 1950s. They titled an article published in the American Journal of Medicine in 1956 ‘Extreme obesity associated with alveolar hypoventilation: a Pickwickian syndrome’. This was in homage to Charles Dickens, whose main character Joe (Fig 2) in The Pickwick Papers (1836) falls asleep in any situation at any time of day. Pickwickian syndrome can still be found in medical dictionaries to this day.
In 1964, a study showed that 91 per cent of snoring patients had a narrow pharynx, elongated soft palate and uvula. This led in due course to tracheostomy being the first successful treatment – despite serious disadvantages, including recurrent purulent bronchitis and speech difficulties.
Sleep apnoea
Pickwickian syndrome is now commonly called obstructive sleep apnoea (OSA) and we should distinguish between the two types – obstructive sleep apnoea and central sleep apnoea (CSA).
OSA is the result of the mechanical collapse of the upper airway, whereas CSA arises from a reduction or lack of brainstem activity regulating the respiratory muscles activity in failing to send a message to the respiratory muscles to breathe. Each type of apnoea is managed differently.
Where does airway collapse occur?
The upper (pharyngeal) airway from the hard palate to the larynx is made up of hard tissues – the hard palate, the maxilla and mandible, the nasal turbinates, the hyoid bone (anteriorly) and cervical vertebrae (posteriorly) and a collapse of the tube in between.
The pharynx has three segments: from top to bottom the nasopharynx, the oropharynx and the hypopharynx.
The nasopharynx connects the nose to the mouth and remains open when surrounding muscles flex so that the person can continue breathing. The salpingopharyngeal fold and tubal tonsils surround it.
The soft palate (velum or muscular palate) separates the nasopharynx from the oropharynx, which extends from the uvula to the level of the hyoid bone. The oropharynx is divided into the retroglossal pharynx (from the soft palate to the epiglottis) and the hypopharynx (from the epiglottis to the larynx). In addition, the area between the retropalatal and retroglossal pharynx is also called the velum (Fig 3).
OSA and bony structures
We’ll look now at what bony structures can predispose to OSA. Any abnormalities in the hard tissues such as the mandible, maxilla, hard palate and hyoid bone can contribute to the displacement of the soft structures, such as the tongue. This, in turn, can lead to airway obstruction. Bony protuberances along the cervical vertebrae can also lead to airway obstruction.
The most common skeletal abnormally likely to lead to OSA is a short mandible. Shiroh Isono et al published in the Journal of Applied Physiology in April 1997 concluded: “The results support the anatomic hypothesis that sleep apneic subjects have a structurally narrowed and collapsible pharynx.”
Studies have also found that the more inferior the hyoid bone, the more likely the tongue will be displaced lower and potentially increase the risk of developing OSA (Fig 4).
OSA and soft tissues
Typically, someone with OSA will have a disproportionately higher volume of soft tissue compared to their hard tissue cage and any inflammation of these soft tissues may contribute to airway obstruction. Whether increased fat pads also predispose to airway compression is disputed.
Nocturnal rostral distribution, whereby fluid is displaced from the legs to the neck during sleep, can lead to increased pressure in the blood vessels, hence the sort of upper airway collapse seen in patients with, for example, heart failure.
OSA and general factors
Here, I shall discuss how gender, ethnicity, age and obesity can contribute to the pathophysiology of OSA.
OSA is present in twice as many men (around 4 per cent of the population) as women (2 per cent). Incidentally, these figures are only for those diagnosed with OSA – probably only one fifth of all those who have the disease.
It appears men are more likely to experience airway collapse because of their higher pharyngeal resistance, but the effect of testosterone may also be a factor. Women’s risk of having sleep apnoea increases after menopause, and those who have the condition have more severe symptoms than do younger women.
Although there has only been limited research, there are ethnic differences in the prevalence and severity of OSA. For instance, epidemiological studies show African-Americans have a larger tongue and longer soft palate, whereas Asians generally have a shorter maxilla and mandible and lower BMI than Caucasians.
A study by Wen Bun Leong et al published in the Journal of Clinical Sleep Medicine (Vol 09, No. 09) concluded: “OSA prevalence and comorbidities was greater in severely obese South Asians compared to obese white Europeans. South Asians also had more severe OSA compared to BMI-matched white Europeans.”
There is an increased risk of OSA as one gets older. Ageing may lead to reduced muscle tone and hence greater likelihood of airway collapse. NHS Choices states that: “Although OSA can occur at any age, it is more common in people who are over 40.”
By contrast, obesity is a significant factor in the development of OSA. This may be due to the increased fat deposits in the neck resulting in greater extraluminal pressure and airway narrowing. Certainly, it has been shown that fat deposition around the neck is more critical than body mass, which is why neck circumference is such an important measure. NHS Choices states: “Men with a collar size greater than around 43cm (17 inches) have an increased risk of developing OSA.”
Other general factors that may contribute to the pathophysiology of OSA include high blood pressure, diabetes, genetics, smoking, alcohol and drugs (hypnotics and sedatives).
So far, so interesting
Before I move on from the physiology to the pathology, I’ll summarise what we’ve learned so far.
For factors contributing to the pathophysiology of obstructive sleep apnoea, I invariably use the table in an article by Deegan and McNicholas published in European Respiratory Journal in 1995. Incidentally, I recommend this article Pathophysiology of obstructive sleep apnoea, because it goes into much greater depth than I have room for here. It is available at http://erj.ersjournals.com/content/8/7/1161
General factors are anthropometric (male sex, age, obesity), drugs (ethanol, hyponotics) and genetics.
Reduced upper airway calibre can be a result of specific anatomical lesions (enlarged tonsils, micrognathia – a lower jaw smaller than normal), neck flexion and nasal obstruction.
Mechanical factors are supine posture, increased upper airways resistance and increased upper airway compliance.
Upper airway muscle function – abnormal upper airways muscle activity; impaired relationship of upper airways muscle and diaphragm contraction.
Upper airway reflexes – impaired response to negative pressure and feedback from the lungs.
Central factors are reduced chemical drives, increased periodicity of central drive and inadequate response to breath loading.
Finally, under the heading of arousal, we have impaired arousal responses and post-apnoeac hyperventilation.
The consequences
Sleep-related breathing disorders (SRBDs) have an adverse effect on the cardiovascular, metabolic, endocrine, nervous and immune systems and the metabolic cycle.
There have been numerous studies on the relationship between SRBDs and cardiovascular consequences. Originally the coexistence of SRBDs with cardiovascular diseases was thought to be due to shared risk factors such as age, gender and obesity. However, Bananian et al published an article ‘Cardiovascular consequences of sleep-related breathing disorders’, in which they stated: “…recent epidemiologic data confirm an independent association between SRBDs and the different manifestations of cardiovascular diseases.” (http://europepmc.org/abstract/med/12350242).
Kathleen A Ferguson and John A Fleetham published an article in Thorax in 1995 which went into great detail about the consequences of sleep disordered breathing (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1021319/).
In relation to cardiac consequences, they cited systemic hypertension, stating: “In patients with sleep disordered breathing, there are brief phasic changes in blood pressure superimposed on a cyclical pattern which coincide with the upper airways obstruction.”
For pulmonary hypertension/right heart failure, they wrote: “The cyclical changes in pulmonary artery pressure parallel the changes in systemic blood pressure.”
They also considered cardiac function – “Treatment of sleep disordered breathing can improve cardiac function in selected patients” – ischaemic heart disease (coronary artery disease) and cardiac arrhythmia (irregular heartbeat).
The Wisconsin Sleep Cohort (WASC) is an ongoing longitudinal study of the causes, consequences and natural history of sleep disorders, particularly sleep apnoea, which has been running for more than 20 years. Research into coronary heart disease as part of WASC, published in SLEEP, the joint publication of the Sleep Research Society and the American Academy of Sleep Medicine, concluded: “Participants with untreated severe sleep disordered breathing (AHI > 30) were 2.6 times more likely to have an incident coronary heart disease or heart failure compared to those without sleep disordered breathing. Our findings support the postulated adverse effects of sleep disordered breathing on coronary heart disease and heart failure.” (see http://www.journalsleep.org/ViewAbstract.aspx?pid=29996).
AHI stands for apnoea-hypopnoea index – the average apnoeas and hypopnoeas that occur during sleep. Mild OSA is in the AHI scale of five to 15. Moderate OSA is when AHI is between 15 to 30. Severe OSA, with an AHI of 30 or more, could, for example, mean falling asleep while driving.
The metabolic syndrome, including cardiovascular disease, diabetes and stroke, is diagnosed when an individual presents with three or more of the following factors:
- Increased waistline with high BMI
- Increased blood cholesterol HDL
- Increased blood triglycerides
- Increased blood pressure
- Impaired fasting glucose.
Other consequences of SRBDs include cerebrovascular disease, excessive daytime sleepiness, personality and behavioural changes, decreased libido and/or impotence and nocturia (the need to urinate during the night).
For clarity and convenience, I list these consequences of SRBDs below:
- Daytime fatigue with an increased risk of road traffic and work-related accidents
- Decreased cognitive function
- Increased risk of cardiovascular disease
- Increased risk of diabetes
- Eye complications
- Memory loss
- Learning difficulty and growth in children
- Sleep-deprived partners with ensuing marital strife
- Complications during sedation and general anaesthetic for surgery
- Morning headaches
- Mood swings and depression
- Nocturia
- Decreased libido and erectile dysfunction
- Depression
- Insomnia.
Conclusion
Snoring and obstructive sleep apnoea are serious conditions with possible grave consequences, including an increased mortality rate. Dentists can work alongside the medical profession to help with the screening, assessment and subsequent treatment process.
The British Society of Dental Sleep Medicine (BSDSM), of which I am president, is a professional organisation for members of the dental team interested in helping patients seeking help for snoring and obstructive sleep apnoea. It advocates medical diagnosis and the provision of the most appropriate therapy.
The BSDSM runs one-day introduction to dental sleep medicine courses and more information, as well as online booking, is at http://www.dentalsleepmed.org.uk
About the Author
Dr Aditi Desai graduated as a dentist from University of Wales, Cardiff in 1977. President of the British Society of Dental Sleep Medicine, she serves on the odontological council and sleep medicine council at the Royal Society of Medicine. Aditi is founder of Global Sleep Solutions, a company set up to bring about a multidisciplinary approach to the management of sleep disorders. Aditi is a restorative dentist with special interest in sleep medicine and part of a multidisciplinary dental specialty team in London’s Harley Street. Working in Harley Street, Portsmouth and London Bridge Hospital as part of the Sleep Service, she maintains a multidisciplinary team approach in her management of patients with sleep disordered breathing. Aditi lectures in the UK and abroad on the subject of dental sleep medicine and is accredited by the European Academy of Dental Sleep Medicine (EADSM).